Unveiling the Balancing Equations Lab Answer Key, this comprehensive guide empowers you to decipher the intricate dance of chemical reactions. Delve into the realm of chemistry, where understanding the art of balancing equations unlocks the mysteries of the molecular world.
Discover the significance of balancing equations in comprehending chemical transformations. Explore step-by-step methods for achieving equilibrium, unravel common pitfalls, and delve into practical applications that solidify your grasp of this fundamental concept.
Understanding Balancing Equations
Balancing chemical equations is a fundamental step in understanding chemical reactions. It involves adjusting the coefficients of reactants and products to ensure that the number of atoms of each element is the same on both sides of the equation. This ensures that the law of conservation of mass is upheld, which states that matter cannot be created or destroyed in a chemical reaction.
Balancing equations is essential for several reasons. First, it allows us to determine the stoichiometry of a reaction, which is the quantitative relationship between the reactants and products. This information is crucial for predicting the amount of reactants and products involved in a reaction and for calculating reaction yields.
Second, balancing equations helps us understand the chemical mechanisms involved in reactions. By examining the balanced equation, we can identify the reactants that are consumed and the products that are formed, as well as the changes that occur to the atoms and molecules involved.
Significance of Balancing Equations
- Ensures the law of conservation of mass is upheld.
- Determines the stoichiometry of a reaction, providing quantitative relationships between reactants and products.
- Helps understand the chemical mechanisms involved in reactions by identifying reactants consumed, products formed, and atomic/molecular changes.
Methods for Balancing Equations
Balancing equations is a fundamental skill in chemistry. It involves adjusting the coefficients in a chemical equation to ensure that the number of atoms of each element is the same on both sides of the equation. There are several methods for balancing equations, each with its own advantages and disadvantages.
Inspection Method
The inspection method is a simple and straightforward method that can be used to balance equations for simple reactions. It involves looking at the equation and adjusting the coefficients until the number of atoms of each element is the same on both sides.
For example, to balance the equation for the combustion of methane:
CH4+ 2O 2→ CO 2+ 2H 2O
We can see that there are one carbon atom, four hydrogen atoms, and two oxygen atoms on the left side of the equation, but only one carbon atom, two hydrogen atoms, and four oxygen atoms on the right side. To balance the equation, we need to adjust the coefficients:
CH4+ 2O 2→ CO2+ 2H 2O
Now, the equation is balanced with one carbon atom, four hydrogen atoms, and four oxygen atoms on both sides.
Examples of Balancing Equations
To illustrate the balancing of chemical equations, let’s explore a few examples. These examples will showcase the initial unbalanced equations and the steps taken to achieve balance.
Table of Unbalanced and Balanced Equations
The following table presents several chemical reactions, their initial unbalanced equations, and their balanced counterparts:
| Unbalanced Equation | Balanced Equation |
|---|---|
| H2 + O2 → H2O | 2H2 + O2 → 2H2O |
| Fe + HCl → FeCl2 + H2 | Fe + 2HCl → FeCl2 + H2 |
| C3H8 + O2 → CO2 + H2O | C3H8 + 5O2 → 3CO2 + 4H2O |
| NH3 + HCl → NH4Cl | NH3 + HCl → NH4Cl |
| 2Na + 2H2O → 2NaOH + H2 | 2Na + 2H2O → 2NaOH + H2 |
In each case, the unbalanced equation is shown in red, highlighting the changes made to achieve balance. Coefficients are adjusted in front of the chemical formulas to ensure that the number of atoms of each element is the same on both sides of the equation.
Balancing Equations in Practice
To solidify understanding of balancing equations, it’s beneficial to engage in hands-on activities or simulations. These practical experiences allow students to apply the concepts they’ve learned and develop a deeper comprehension of the process.
Here’s a lab activity that demonstrates the process of balancing equations:
Materials
- Chemical formulas for reactants and products
- Index cards or paper
- Markers or pens
Procedure
- Write the unbalanced equation on an index card or paper.
- Divide the index card into two columns, one for reactants and one for products.
- For each element in the reactants, count the number of atoms and write it as a subscript after the element symbol.
- Repeat step 3 for the products.
- Adjust the coefficients in front of each formula until the number of atoms for each element is the same on both sides of the equation.
- Check the equation to ensure it is balanced.
Observations, Balancing equations lab answer key
- Students will observe that the number of atoms for each element is the same on both sides of the balanced equation.
- They will also notice that the coefficients in front of the formulas represent the number of molecules of each reactant and product involved in the reaction.
Common Errors in Balancing Equations
Balancing equations is a fundamental skill in chemistry. However, it is not without its challenges. Several common mistakes can arise when attempting to balance equations. Understanding these errors and their causes can help you avoid them and improve your accuracy in balancing equations.
Incorrect Coefficients
One of the most common errors is using incorrect coefficients. Coefficients are the numbers placed in front of chemical formulas to balance the number of atoms on both sides of the equation. Incorrect coefficients can lead to an unbalanced equation that does not accurately represent the chemical reaction.
To avoid this error, carefully count the number of atoms of each element on both sides of the equation. Ensure that the number of atoms is the same on both sides before adjusting the coefficients.
Ignoring Spectators
Spectator ions are ions that appear on both sides of a chemical equation and do not participate in the reaction. Neglecting spectator ions when balancing equations can lead to incorrect coefficients for the reactants and products.
To avoid this error, identify the spectator ions by examining the chemical formulas. The spectator ions should be excluded when counting the atoms to determine the coefficients.
Changing the Chemical Formulas
Another common error is changing the chemical formulas of the reactants or products to balance the equation. This is incorrect and can lead to an inaccurate representation of the chemical reaction.
To avoid this error, always maintain the original chemical formulas of the reactants and products. If you find that you need to change a chemical formula to balance the equation, it is likely that you have made another error.
Forgetting to Balance Elements One at a Time
When balancing equations with multiple elements, it is important to balance the elements one at a time. Balancing all the elements simultaneously can lead to confusion and errors.
To avoid this error, focus on balancing one element at a time. Once that element is balanced, move on to the next element. Repeat this process until all the elements are balanced.
FAQ Guide: Balancing Equations Lab Answer Key
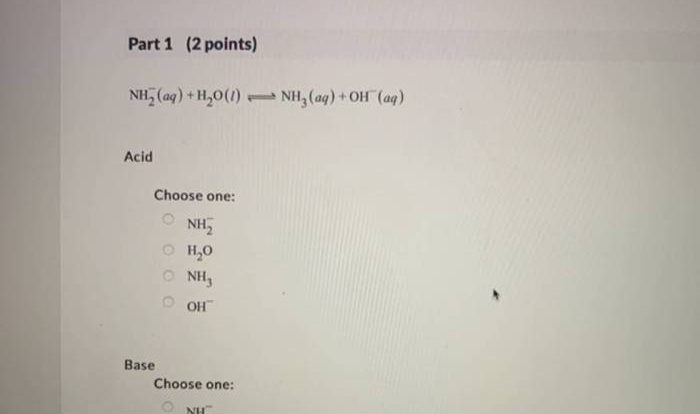
What is the significance of balancing equations in chemistry?
Balancing equations ensures that the number of atoms of each element on the reactants’ side matches the number of atoms of the same element on the products’ side. This reflects the law of conservation of mass, a fundamental principle in chemical reactions.
How do I balance equations using the half-reaction method?
The half-reaction method involves splitting the overall reaction into two half-reactions, one for oxidation and one for reduction. Each half-reaction is balanced separately before combining them to obtain the balanced overall equation.
What are some common errors to avoid when balancing equations?
Common errors include changing the chemical formulas of reactants or products, altering the coefficients in front of the species, and neglecting to balance all elements in the equation.